‘It’s a Minefield’: Biden Health Pick Must Tread Carefully on Abortion and Family Planning
President Biden vowed to reverse reproductive health restrictions enacted by President Trump. His pick to run HHS, Xavier Becerra, fought the Trump efforts but must now navigate a difficult legal and political landscape.
New Single-Payer Bill Intensifies Newsom’s Political Peril
With the introduction of a single-payer bill Friday, a group of California Democratic lawmakers set the terms of the health care debate in the Capitol this year. The move puts Gov. Gavin Newsom in a delicate political position, threatening to alienate voters as he faces a likely recall election.
Vaccines Go Mobile to Keep Seniors From Slipping Through the Cracks
A strike team of nurses and others is vaccinating Contra Costa County’s hardest-hit populations right where they live.
CVS and Walgreens Under Fire for Slow Pace of Vaccination in Nursing Homes
A federal program that sends retail pharmacists into nursing homes to vaccinate residents and workers has been hindered by bureaucratic hurdles and scheduling woes.
Agrícolas, bomberos y azafatas buscan estar entre los primeros en recibir la vacuna
Trabajadores de salud de primera línea, y residentes y personal de hogares de adultos mayores, recibirán las dosis de la vacuna contra COVID primero, pero… ¿quiénes le seguirán?
Farmworkers, Firefighters and Flight Attendants Jockey for Vaccine Priority
Everyone — from toilet paper manufacturers to patient advocates — is lobbying state advisory boards, arguing their members are essential, vulnerable or both — and, thus, most deserving of an early vaccine.
Limpieza dental, gafas nuevas… y la vacuna contra COVID-19
Con múltiples vacunas de COVID-19 en camino, optometristas y dentistas presionan para que se autorice vacunar a los pacientes durante los exámenes oculares de rutina y las limpiezas dentales.
Come for Your Eye Exam, Leave With a Band-Aid on Your Arm
Dentists and optometrists across the country are trying to join in the fight to get everyone vaccinated against COVID-19, the flu and other diseases.
App-Based Companies Pushing Prop. 22 Say Drivers Will Get Health Benefits. Will They?
Ride-sharing and delivery services such as Uber, Lyft, DoorDash and Instacart are bankrolling California’s Proposition 22, which would keep their drivers classified as independent contractors, not employees. But health benefits? That’s something of a stretch.
Californians Asked to Pony Up for Stem Cell Research — Again
More than a decade of research tied to California’s stem cell agency hasn’t yielded many cures or much revenue. But backers of a ballot initiative that asks voters for billions more in funding say the work is vital for patients and the scientific community.
Pandemia obstaculiza al preciado bloque de votantes en centros de adultos mayores
Muchos seniors que necesitan ayuda para obtener o llenar sus boletas podrían sufrir la consecuencias por el cambio de las reglas sobre visitas familiares.
Pandemic Erects Barriers for Prized Bloc of Voters in Nursing Homes, Senior Facilities
Voting is a point of pride for many older Americans, and senior living facilities in past years have encouraged the civic act by hosting voting precincts, providing transportation to the polls and bringing in groups to help explain election issues. But fears of the spread of the coronavirus among this vulnerable population make voting more difficult this year.
‘Terrible Role-Modeling’: California Lawmakers Flout Pandemic Etiquette
As California workers and schoolchildren struggled to work from home, state lawmakers met in person. And as their legislative session came to a close in late August, they broke COVID rules: They huddled, let their masks slip below their noses, removed their masks to drink coffee — and required a new mom to vote in person while toting her hungry newborn.
In Legislative Shuffle, California Prioritizes Safety Gear and Sick Leave During Crisis
Lawmakers are calling on Gov. Gavin Newsom to sign bills that would address the challenges of the current COVID-19 crisis and help the state prepare for future pandemics.
Long-Fought Nurse Practitioner Independence Bill Heads to Newsom
The measure caps one of the most contentious health policy debates in recent memory, potentially altering how Californians get their medical care. Gov. Gavin Newsom has until the end of September to sign or veto it.
Amid COVID Chaos, California Legislators Fight for Major Health Care Bills
There’s less time, less attention and fewer resources this year, but that isn’t stopping lawmakers from acting on controversial health care legislation not directly related to the coronavirus pandemic.
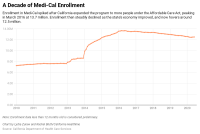
Medicaid Mystery: Millions of Enrollees Haven’t Materialized in California
State officials had projected that 2 million Californians would join Medi-Cal, the state’s health insurance program for low-income people, by July because of the economic devastation wrought by COVID-19. Yet enrollment has barely budged, and why is unclear.
A pesar del aumento de casos, California frena fondos multimillonarios para pruebas de COVID
El estado ya no financiará nuevos sitios de prueba, a pesar de las súplicas de los condados para obtener asistencia adicional. También ha cerrado algunos espacios y los ha trasladado a otros lugares.
As Cases Spike, California Pauses Multimillion-Dollar Testing Expansion
California is cutting off funding for COVID-19 testing just when counties say they need more resources in rural and disadvantaged areas.
Easy-breezy guest writer Rachel Bluth fills you in on a healthy dose of news from this past week.