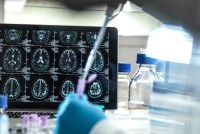
Amgen Plows Ahead With Costly, Highly Toxic Cancer Dosing Despite FDA Challenge
The FDA told Amgen to test whether a quarter-dose of its lung cancer drug worked as well as the amount recommended on the product label. It did and with fewer side effects. But Amgen is sticking to the higher dose — which earns it an additional $180,000 a year per patient.
Una prueba genética podría salvar la vida de cientos de pacientes en quimioterapia
Estos tipos de quimioterapia comunes son difíciles de tolerar en general, pero para los pacientes que tienen deficiencia de una enzima que metaboliza la droga, puede ser una tortura o causar la muerte.
Overdosing on Chemo: A Common Gene Test Could Save Hundreds of Lives Each Year
The FDA and some oncologists have resisted efforts to require a quick, cheap gene test that could prevent thousands of deaths from a bad reaction to a common cancer drug.
Why Covid Patients Who Could Most Benefit From Paxlovid Still Aren’t Getting It
Price worries, bureaucratic obstacles, and “I’m-over-covid-itis” slow uptake of a drug that’s complicated to take but often effective.
Patients See First Savings From Biden’s Drug Price Push, as Pharma Lines Up Its Lawyers
A restructuring of the Medicare drug benefit has wiped out big drug bills for people who need expensive medicines. But the legal battle over drug negotiations means uncertainty over long-term savings.
What the Health Care Sector Was Selling at the J.P. Morgan Confab
When bankers and investors flocked to San Francisco for the largest gathering of health care industry investors, the buzz was all about artificial intelligence, the next hit weight-loss drug, and new opportunities to make money through nonprofit hospitals.
A New Test Could Save Arthritis Patients Time, Money, and Pain. But Will It Be Used?
Stories of chronic pain, drug-hopping, and insurance meddling are all too common among patients with rheumatoid arthritis. Precision medicine offers new hope.
The Market for Biosimilars Is Funky. The Industry Thinks PBMs Are To Blame
Over the past year there’s been movement to rein in the three big PBMs, which face little regulation though they help set drug prices and drug choices for 80 percent of Americans and their doctors. The House voted Dec. 11, 320-71, for legislation that would require the PBMs to change some of the ways they […]
Biden Administration’s Limit on Drug Industry Middlemen Backfires, Pharmacists Say
A rule taking effect Jan. 1 was intended to stop one set of abuses by pharmacy benefit managers, or PBMs, but some pharmacists say it’s enabling these price brokers to simply do new things unfairly.
Humira lleva 20 años disfrutando de una exclusividad muy cara en el país. Sus competidores podrían ahorrarle al sistema sanitario $9,000 millones.
Save Billions or Stick With Humira? Drug Brokers Steer Americans to the Costly Choice
Thousands of patients with autoimmune diseases who rely on Humira, with a list price of $6,600 a month, could get financial relief from new low-cost rivals. So far, the pharmacy benefit managers that control drug prices in America have not delivered on those savings.
Por qué los CDC recomiendan el nuevo refuerzo contra covid para todos
El Comité Asesor sobre Prácticas de Inmunización de los CDC votó 13-1 a favor de la moción después de meses de debate sobre si limitar los refuerzos a grupos de alto riesgo.
Why the CDC Has Recommended New Covid Boosters for All
As covid-19 hospitalizations tick upward with fall approaching, the CDC says it’s time for new boosters — and not only for those at highest risk of serious disease. Here are seven things you need to know.
A pesar de las amplias recomendaciones para el refuerzo anticovid actualizado publicadas el otoño pasado, sólo el 17% de la población la recibió, y alrededor del 43% de las personas de 65 años o más.
Pfizer and Moderna Are Pushing the New Covid Booster. Should You Get It? The CDC Is About to Decide.
Chances are, if you aren’t older, chronically ill, or obese, you don’t need a forthcoming covid vaccine to stay out of the hospital. But it probably wouldn’t hurt.
5 Things to Know About the New Drug Pricing Negotiations
The Biden administration unveiled the first 10 drugs subject to price negotiations, taking a swipe at the pharmaceutical industry. But what does it mean for patients?
The Real Costs of the New Alzheimer’s Drug, Most of Which Will Fall to Taxpayers
The annual cost of lecanemab treatment quadruples if the expense of brain scans to monitor for bleeds and other associated care is factored in. The full financial toll likely puts it beyond reach for low-income seniors at risk of Alzheimer’s, experts say.
What You Need to Know About the Drug Price Fight in Those TV Ads
At least nine bills introduced in Congress take aim at pharmacy benefit managers, the powerful middlemen that channel prescription drugs to patients.
Why the Next Big Hope for Alzheimer’s Might Not Help Most Black Patients
Black patients and other minorities tend to be diagnosed at later stages of the disease, which would exclude them from use of Leqembi. Few Black people were included in the main trial of the drug.
El cisplatino y el carboplatino son algunos de los medicamentos que escasean, así como otros 12 contra el cáncer, pastillas para el trastorno por déficit de atención con hiperactividad, anticoagulantes y antibióticos.