Congress Likely to Kick the Can on Covid-Era Telehealth Policies
With an end-of-year deadline and a presidential election approaching, payment rules that fueled rapid expansion of telehealth in the United States face a last-minute congressional decision.
End of Internet Subsidies for Low-Income Households Threatens Telehealth Access
A federal program that helped pay for more than 23 million low-income households’ internet access runs out of money soon. The end of the subsidy launched earlier in the pandemic could have profound impacts on health care access.
Medicare Advantage Is Popular, but Some Beneficiaries Feel Buyer’s Remorse
Medicare Advantage plans are booming — 30.8 million of the 60 million Americans with Medicare are now enrolled in the private plans rather than the traditional government-run program. But a little-known fact: Once you’re in a Medicare Advantage plan, you may not be able to get out. Traditional Medicare usually requires beneficiaries to pay 20 […]
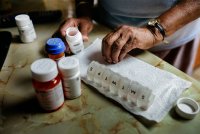
Watch: Older Americans Say They Feel Stuck in Medicare Advantage Plans
You’ve probably seen advertising about Medicare Advantage plans. KFF Health News’ Sarah Jane Tribble explains the pros and cons of this insurance option as enrollment in these plans increases.
Federal Program to Save Rural Hospitals Feels ‘Growing Pains’
Fewer than two dozen rural hospitals were converted into Rural Emergency Hospitals in the program’s first year. Now, advocates and lawmakers say tweaks to the law are necessary to lure more takers and keep health care in rural communities.
Adultos mayores se sienten “atrapados” en planes de Medicare Advantage
Al parecer el programa de planes privados para adultos mayores comienza a presentar obstáculos cuando surgen enfermedades.
Older Americans Say They Feel Trapped in Medicare Advantage Plans
As enrollment in private Medicare Advantage plans grows, so do concerns about how well the insurance works, including from those who say they have become trapped in the private plans as their health declines.
¿Pueden los médicos de familia salvar a las zonas rurales de la crisis de obstetras?
El número de bebés que murieron antes de cumplir su primer año aumentó el año pasado; y más de la mitad de los condados rurales no tienen servicios hospitalarios para partos.
Can Family Doctors Deliver Rural America From Its Maternal Health Crisis?
Family medicine doctors already deliver most of rural America’s babies, and efforts to train more in obstetrics care are seen as a way to cope with labor and delivery unit closures.
Tiny, Rural Hospitals Feel the Pinch as Medicare Advantage Plans Grow
More than half of seniors are enrolled in private Medicare Advantage plans instead of traditional Medicare. Rural enrollment has increased fourfold and many small-town hospitals say that threatens their viability.
What Mobile Clinics in Dollar General Parking Lots Say About Health Care in Rural America
Dollar General’s pilot mobile clinic program has been touted by company officials, rural health experts, and analysts as a model that could help solve rural America’s primary care shortage. But its Tennessee launch has been met with local skepticism.
Facing Criticism, Feds Award First Maternal Health Grant to a Predominantly Black Rural Area
Mississippi has the highest rate of maternal mortality in the U.S. Now, it also has a federal grant to help in rural areas. The award could signal more flexibility from federal officials.
Black, Rural Southern Women at Gravest Risk From Pregnancy Miss Out on Maternal Health Aid
A federal program meant to reduce maternal and infant mortality in rural areas isn’t reaching Black women who are most likely to die from pregnancy-related causes.
He Returned to the US for His Daughter’s Wedding. He Left With a $42,000 Hospital Bill.
After emergency surgery, an American expatriate with Swiss insurance now carries the baggage of a five-figure bill. Costs for medical care in the U.S. can be two to three times the rates in other developed countries, so foreigners and expats with good insurance in their home countries need travel insurance to protect themselves from “crazy prices.”
New Mexico Program to Reduce Maternity Care Deserts in Rural Areas Fights for Survival
A federally funded program in remote New Mexico has helped hundreds of pregnant mothers stay healthy, but it’s running out of time and money despite a growing national maternity care crisis. The four-year, nearly $3 million grant has provided telehealth, coordinated care, and social services to mothers in need.
Congressman Seeks to Plug ‘Shocking Loophole’ Exposed by KHN Investigation
A federal lawmaker has introduced a House bill that would close one of a laundry list of oversight gaps revealed in a recent KHN investigation of the system regulators use to ban fraudsters from billing government health programs, including Medicare and Medicaid.
Legal Questions, Inquiries Intensify Around Noble Health’s Rural Missouri Hospital Closures
A year after private equity-backed Noble Health shuttered two rural Missouri hospitals, a slew of lawsuits and state and federal investigations grind forward. Missouri Attorney General Andrew Bailey recently confirmed an “ongoing” investigation as former employees continue to go unpaid and cope with unpaid medical claims.
Hospitales rurales aplican a nuevo programa federal para intentar sobrevivir
Más de 140 hospitales rurales han cerrado en todo el país desde 2010, y observadores de políticas de salud no están seguros de cuántas de las más de 1,700 instalaciones rurales elegibles para la nueva designación aplicarán a un nuevo programa.
Struggling to Survive, the First Rural Hospitals Line Up for New Federal Lifeline
Hospitals in New Mexico, Texas, and Oklahoma are among the first to apply for a new rural hospital payment model that shifts the focus of services away from overnight stays to outpatient and emergency care. Still, experts say the law needs to be amended to provide the right mix of care for rural communities.
A Bitter Battle Over the ‘Orphan Drug’ Program Leaves Patients’ Pocketbooks at Risk
Patients who depend upon special drugs to treat rare diseases are caught in the crossfire as drugmakers and the FDA battle over regulations that reward companies for developing treatments for relatively small pools of patients.