Latest KFF Health News Stories
FDA Announces Recall of Heart Pumps Linked to Deaths and Injuries
Some pumps used in end-stage heart failure caused a buildup of biological material that blocks blood flow from the device to the heart’s aorta. The FDA’s recall affects nearly 14,000 devices.
Ten Doctors on FDA Panel Reviewing Abbott Heart Device Had Financial Ties With Company
Most of the doctors the FDA tapped to advise it on an Abbott medical device had financial ties to the company. The FDA didn’t disclose the payments.
‘AGGA’ Inventor Testifies His Dental Device Was Not Meant for TMJ or Sleep Apnea
The FDA and Department of Justice are investigating the Anterior Growth Guidance Appliance, or “AGGA.” TMJ and sleep apnea patients have filed lawsuits alleging the device harmed them. Its inventor now says the AGGA was never meant for these ailments.
Deep Flaws in FDA Oversight of Medical Devices, and Patient Harm, Exposed in Lawsuits and Records
Thousands of medical devices are sold, and even implanted, with no safety tests.
Patients Facing Death Are Opting for a Lifesaving Heart Device — But at What Risk?
The HeartMate 3 is considered the safest mechanical heart pump of its kind, but a federal database contains more than 4,500 reports in which the medical device may have caused or contributed to a patient’s death.
Patients Expected Profemur Artificial Hips to Last. Then They Snapped in Half.
The FDA and the manufacturer were alerted to Profemur titanium hips breaking inside U.S. patients as of 2005. It took 15 years to recall the devices. Many fractures could have been avoided.
Thousands Got Exactech Knee or Hip Replacements. Then, Patients Say, the Parts Began to Fail.
In a torrent of lawsuits, patients accuse Florida device maker Exactech of hiding knee and hip implant defects for years. The company denies the allegations.
Feds Launch Criminal Investigation Into ‘AGGA’ Dental Device and Its Inventor
KFF Health News and CBS News recently reported that multiple lawsuits allege the device has led to grievous injuries to patients’ mouths, resulting in loss of teeth.
During In-Flight Emergencies, Sometimes Airlines’ Medical Kits Fall Short
U.S. airlines have response plans for passengers who run into health issues in flight, but planes carry limited and sometimes incomplete medical supplies that can put travelers at risk.
A Shortfall of ECMO Treatment Cost Lives During the Delta Surge
About 50% of the covid-19 patients who got the last-ditch life support treatment at Vanderbilt University Medical Center died. Researchers wanted to know what happened to the many patients they had to turn away because ECMO (extracorporeal membrane oxygenation) machines and the specialized staffers needed were in short supply. The grim answer: 90% of those turned away perished.
Apple, Bose and Others Pump Up the Volume on Hearing Aid Options, Filling Void Left by FDA
A 2017 law designed to help lower the cost of hearing aids mandated that federal officials set rules for a new class of devices consumers could buy without needing to see an audiologist. But those regulations are still on hold.
Medical Device Failures Brought To Light Now Bolster Lawsuits And Research
Millions of injuries and malfunctions once funneled into a hidden Food and Drug Administration database are now available.
Atracción en hospitales: pruebas gratuitas para detectar hernias, ¿funcionan?
Según los Centros para el Control y Prevención de Enfermedades (CDC), en el país se diagnostican aproximadamente 1,6 millones de hernias en la ingle; y se tratan quirúrgicamente 500,000 al año.
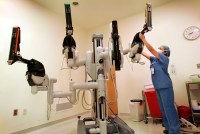
Robotic Surgical Tool, Not Medical Evidence, Drives Free Hernia Screenings
Hospitals around the country are promoting free hernia screenings that tout their robotic surgery tools. But some experts warn such screenings could lead people to get potentially harmful operations that they don’t need.
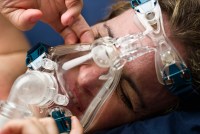
I’m A CPAP Dropout: Why Many Lose Sleep Over Apnea Treatment
An estimated 18 million American adults have sleep apnea. The go-to treatment — a CPAP machine — offers a healthy restful night’s sleep, but many people struggle to use it. As many as 50% of patients stop using the device.
‘An Arm And A Leg’: Journalist Learns The Hard Way That CPAP Compliance Pays
Check the fine print. When you get a prescription for expensive medical equipment, you may need to follow the doctor’s orders — to the letter — to get your health insurance company to pay up.
KHN filed multiple Freedom of Information Act requests over months, and the FDA responded Wednesday saying the data about device malfunctions is now publicly available online.
Five Things We Found In The FDA’s Hidden Device Database
The Food and Drug Administration released two decades of previously hidden data containing millions of injuries or malfunctions by medical devices. Here’s what we’ve learned so far.
More Than Half Of Surgical Stapler Malfunctions Went To Hidden FDA Database
The FDA reveals that 56,000 malfunctions associated with surgical staplers weren’t reported through its traditional public reporting system.
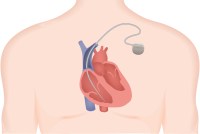
Hidden Reports Masked The Scope Of Widespread Harm From Faulty Heart Device
The Food and Drug Administration allowed one company to send 50,000 reports of harm or malfunctions to an internal database even as patients worried about faulty defibrillators lodged in their hearts.