Latest KFF Health News Stories
Sprained Your Ankle? The Cost Of A Brace Could Sprain Your Wallet.
Your health insurance might not cover items such as wheelchairs, walkers, crutches and braces, or you may have to deal with a supplier that has a contract with your insurer.
Study: Nearly Three-Quarters Of Commonly Used Medical Scopes Tainted By Bacteria
The ‘scary’ findings show a discouraging lack of progress in cleaning the devices, despite more vigorous efforts in the wake of deadly superbug outbreaks, experts say.
Judge Orders New Olympus Trial Over Superbug Death
The Seattle jurist finds that Olympus Corp. failed to properly disclose evidence that it knew of concerns about cleaning problems with its redesigned medical scopes years before they hit the market and were linked to dozens of deaths. The company maintains the devices were not defective and intends to appeal.
Heart Device Failure: Medicare Spent $1.5B Over 10 Years To Replace Defective Implants
The inspector general at Health and Human Services says defective pacemakers or defibrillators had to be replaced from 2005 through 2014, costing Medicare $1.5 billion.
To Wage War On Superbugs, FDA Clears Way For Scope With A Disposable Piece
Agency says a removable cap will lower the risk of antibiotic resistant infections but some experts see it as a modest step in curbing the sort of deadly outbreaks that occurred a few years ago.
Quiz: How Well Are You Paying Attention?
To strengthen your core knowledge of health care policy, it helps to be a regular reader of Kaiser Health News. Here’s a pop quiz to gauge what you have learned.
Scope Maker Olympus Hit With $6.6 Million Verdict In Superbug Outbreak Case
In the first case of its kind in the U.S., the company was ordered to pay damages to the hospital where a patient died of an infection linked to a contaminated scope. But jurors also found the hospital negligent, and it was ordered to pay the patients’ family $1 million.
DNA Links Deadly Germs, Tainted Heart Surgery Devices To German Factory
LivaNova plant in Germany is the likely source behind outbreak that has sickened more than 100 people since 2013.
Widow Unleashes Court Fight Against Scope Maker Olympus Over Superbug Outbreak
The Seattle case, the first to reach trial in the U.S., offers possible glimpse into fate of some two dozen lawsuits against manufacturing giant Olympus, accused of failing to address scope contamination linked to numerous deaths. The company faults poor hospital cleaning practices.
Dispositivos que salvan vidas en los Estados Unidos se fabrican en México
Casi todos los estadounidenses con marcapasos -y personas en todo el mundo- caminan portando partes fabricadas en Tijuana, México, en donde se ha creado una industria que podría estar en riesgo si la administración Trump cambia el juego del comercio global.
Also Made In Mexico: Lifesaving Devices
The medical supply industry makes a particularly revelatory case study of the difficulties of untangling global trade.
Renewed Cleaning Efforts For Scopes Not Enough To Vanquish Bacteria
A new study, though small, finds extensive damage to commonly used medical scopes that could trap dangerous bacteria. That raises concerns about the potential for more outbreaks.
Cheaper Over-The-Counter Hearing Aids Could Be On The Way
The FDA and other agencies are loosening restrictions on hearing aid sales and opening the door to less expensive, over-the-counter products.
Grilled About Deadly Superbug Outbreaks, Execs At Scope Maker Olympus Take Fifth
Lawyers who deposed top company officials in a civil case say they declined to answer questions about their failure to warn American hospitals of infection risks. Industry giant Olympus also is the subject of a criminal probe.
The Throwaway Scope: A Way To Ditch Superbugs?
Small manufacturers are betting that disposable medical scopes will slash the risk of infection during procedures. Some doctors are skeptical of the cheaper models.
Deadly Infections Linked To Heart Surgery Device Highlight Holes In FDA Monitoring
The federal agency took 14 months to warn the public about the potential for infections. Officials say they acted as fast as they could.
Bad Hombres, Russian Hackers And … A Medical Device Tax?
Why an obscure revenue raiser for the Affordable Care Act has found its way into a number of congressional campaign ads.
FDA Faults 12 Hospitals For Failing To Disclose Injuries, Deaths Linked To Medical Devices
The agency found several prominent facilities had not followed rules on reporting incidents in which patients were harmed.
California Man Dies After Apparent Failure Of Artificial Heart Compressor
The FDA confirms it is looking into more than one problem with the compressor, which is used to power patients’ artificial hearts.
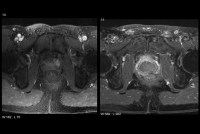
Pricey New Treatment Roils Issues Of How To Treat Prostate Cancer
High-intensity focused ultrasound, often not covered by insurance, leads to discussions about which patients benefit in the real world.