‘We Ain’t Gonna Get It’: Why Bernie Sanders Says His ‘Medicare for All’ Dream Must Wait
As he takes the reins of the Senate Health, Education, Labor & Pensions Committee, the independent from Vermont and implacable champion of “Medicare for All” maps out his strategy for negotiating with Republicans — and Big Pharma.
Por un tecnicismo, niños necesitados podrían no tener acceso a vacunas contra el VRS
El virus respiratorio sincitial afecta a bebés de todas las clases sociales, pero tiende a perjudicar más a los hogares pobres y hacinados
A Technicality Could Keep RSV Shots From Kids in Need
The Vaccines for Children program, which buys more than half the pediatric vaccines in the U.S., may not cover the RSV shot for babies because it’s not technically a vaccine.
FDA Experts Are Still Puzzled Over Who Should Get Which Covid Shots and When
A single booster seems to prevent death and hospitalization in most people, but protection from the current vaccines wanes within months. FDA experts say they need to know more from the Centers for Disease Control and Prevention to decide the best long-term strategy.
Florida Gov. DeSantis Falsely Claims Bivalent Booster Boosts Chances of Covid Infection
Experts say the Florida governor’s conclusion could not be drawn from the study he cited, adding that the research focused on health care workers, who are likelier to be exposed to covid and more likely to be vaccinated. Those findings should not be applied to the general public.
Latino Teens Are Deputized as Health Educators to Sway the Unvaccinated
Some community health groups are training Latino teens to conduct outreach and education, particularly in places where covid vaccine fears linger.
Adolescentes latinos se entrenan para educar sobre las vacunas contra covid
Organizaciones comunitarias de salud en California y en todo el país forman a adolescentes, muchos de ellos latinos, para que actúen como educadores de la salud en la escuela, en las redes sociales y en las comunidades donde persiste el miedo a la vacuna contra covid.
Public Health Agencies Try to Restore Trust as They Fight Misinformation
As public health departments work on improving their message, the skepticism and mistrust often reserved for covid-19 vaccines now threaten other public health priorities, including flu shots and childhood vaccines.
KHN’s ‘What the Health?’: Health Spending? Only Congress Knows
Top negotiators in Congress have agreed to a framework for government spending into next year, but there are details to iron out before a vote — such as the scheduled Medicare payment cuts that have providers worried. Also, the Biden administration reopens its program allowing Americans to request free covid-19 home tests, as hopes for pandemic preparedness measures from Congress dim. Rachel Cohrs of Stat, Alice Miranda Ollstein of Politico, and Rebecca Adams of KHN join KHN’s Mary Agnes Carey to discuss these topics and more. Plus, for extra credit, the panelists recommend their favorite health policy stories of the week they think you should read, too.
Readers and Tweeters Decry Medical Billing Errors, Price-Gouging, and Barriers to Benefits
KHN gives readers a chance to comment on a recent batch of stories.
Blackfeet Nation Challenges Montana Ban on Vaccine Mandates as Infringement on Sovereignty
The Montana tribe has entered a legal fight over whether the state has the right to enforce a prohibition of vaccine mandates on its reservation.
Pfizer’s Covid Cash Powers a ‘Marketing Machine’ on the Hunt for New Supernovas
While sales of its covid vaccines are falling, Pfizer plans to triple the price of the shots and use its bonanza from government contracts to buy and develop new blockbusters.
KHN’s ‘What the Health?’: Voters Will Get Their Say on Multiple Health Issues
Abortion isn’t the only health issue voters will be asked to decide in state ballot questions next month. Proposals about medical debt, Medicaid expansion, and whether health care should be a right are on ballots in various states. Meanwhile, the latest lawsuit challenging the Affordable Care Act has expanded to cover all preventive care. Alice Miranda Ollstein of Politico, Jessie Hellmann of CQ Roll Call, and Victoria Knight of Axios join KHN’s Julie Rovner to discuss these topics and more.
El vínculo con la esclerosis múltiple impulsa una vacuna contra el virus Epstein-Barr
Los científicos llevan años intentando desarrollar vacunas contra este virus. Sin embargo, recientemente varios avances en la investigación médica han dado más urgencia a la búsqueda y más esperanzas de éxito.
As Links to MS Deepen, Researchers Accelerate Efforts to Develop an Epstein-Barr Vaccine
Recent leaps in medical research have lent urgency to the quest to develop a vaccine against Epstein-Barr, a ubiquitous virus that has been linked to a range of illnesses, from mononucleosis to multiple sclerosis and several cancers.
Will Covid Spike Again This Fall? 6 Tips to Help You Stay Safe
Recent research suggests that the covid virus is mutating to better dodge people’s immune defenses. It could soon evade monoclonal antibodies used to treat covid. KHN examines what public health officials believe is on the horizon and how best to fight the disease.
Watch: What Experts Advise for Seniors Living Under the Long Shadow of Covid
For older people, the pandemic is as taxing and worrisome as ever. Experts in geriatric care, mental health, social services, and infectious disease joined a KHN-Hartford Foundation panel to talk about a third covid winter and its outsize toll on seniors.
Watch: Meet the Latest Fact-Checker — Your Doctor
KHN’s Mary Agnes Carey talks with American Medical Association President Dr. Jack Resneck Jr. about how misinformation affects doctors and their daily efforts to treat patients.
Is Covid ‘Under Control’ in the US? Experts Say Yes
PolitiFact has been tracking this campaign promise since 2020. Experts are now saying it’s fair to describe the covid pandemic as “under control.”
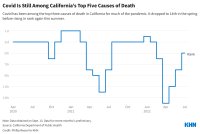
Covid Still Kills, but the Demographics of Its Victims Are Shifting
Californians were far less likely to die from covid in the first seven months of 2022 than during the first two years of the pandemic. Still, the virus remained among the state’s leading causes of death in July, outpacing diabetes, accidental death, and a host of debilitating diseases. We break down who’s at risk.