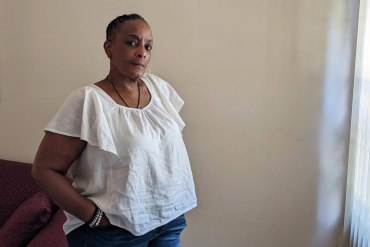Biden Administration to Ban Medical Debt From Americans’ Credit Scores
The White House said the Consumer Financial Protection Bureau will develop new regulations that would prevent unpaid medical bills from being counted on credit reports.
Black, Rural Southern Women at Gravest Risk From Pregnancy Miss Out on Maternal Health Aid
A federal program meant to reduce maternal and infant mortality in rural areas isn’t reaching Black women who are most likely to die from pregnancy-related causes.
FDA Evaluates ‘Safety Concerns’ Over Dental Devices Featured in KHN-CBS Investigation
A KHN and CBS News investigation found that a dental appliance called the AGGA has been used by more than 10,000 patients, and multiple lawsuits allege it has caused grievous harm to patients.
Congressman Seeks to Plug ‘Shocking Loophole’ Exposed by KHN Investigation
A federal lawmaker has introduced a House bill that would close one of a laundry list of oversight gaps revealed in a recent KHN investigation of the system regulators use to ban fraudsters from billing government health programs, including Medicare and Medicaid.
$2,700 Ambulance Bill Pulled Back From Collections
After reporting from KHN, NPR, and CBS News, a patient’s $2,700 ambulance bill was pulled back from collections.
Montana Health Officials Aim to Boost Oversight of Nonprofit Hospitals’ Giving
Montana is one of the latest states seeking to increase oversight of nonprofit hospitals’ giving to ensure they justify their tax-exempt status.
Georgia Bill Aims to Limit Profits of Medicaid Managed-Care Companies
Georgia lawmakers unveiled a mental health bill that would limit the profits of the managed-care companies that serve Medicaid patients. KHN previously reported that Georgia, unlike most states, does not set a medical loss ratio for the companies’ spending on medical care and quality improvements.
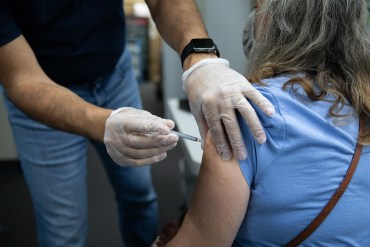
CDC Tells Pharmacies to Give 4th Covid Shots to Immunocompromised Patients
The health agency and the White House acted in the wake of a KHN story about pharmacists refusing to give shots to patients with moderate to severe immune suppression.
NY Reaches Agreement With DOJ Over Vaccine Access for Blind People
Following a February KHN investigation into covid vaccine accessibility, the Department of Justice reached an agreement with five New York government agencies to make their websites accessible to people who are visually impaired.
Congress Cites KHN Investigation in Probe of National Academies
The House oversight committee is requesting conflict-of-interest disclosure forms from a National Academies committee studying organ transplants. KHN previously reported on apparent conflicts among members of a committee studying drug waste.
Labor Department Issues Emergency Rules to Protect Health Care Workers From Covid
Citing the deaths of thousands of health care workers, the new rules will force employers to report fatalities or hospitalizations to the Occupational Safety and Health Administration, and provide higher-quality protective gear, among other actions.
In Reversal, Kansas Will Count All Positive COVID Cases, Even Asymptomatic Ones
Following a KCUR report, Kansas officials said the state’s public reporting of pandemic trends will count all tests that come back positive for the new coronavirus, even when the patient has no symptoms.
‘Red Dawn Breaking Bad’: Officials Warned About Safety Gear Shortfall Early On, Emails Show
As President Donald Trump called the nation “in good shape” to handle COVID-19, a cache of emails released by officials in Washington state show that top public health authorities feared gear shortages and doctor safety in the early epicenter of sickness and deaths.
Reports Of Patients’ Deaths Linked To Heart Devices Lurk Below Radar
Because of a little-known federal exemption program, death data about heart devices sits in inaccessible FDA files that can take up to two years for the public to see under open-records laws.
Paying It Forward: ‘Bill Of The Month’ Series, A Vital Toolkit For Patients, Wraps Year 2
In our ongoing, crowdsourced investigation with NPR and CBS, we’ve armed future health system pilgrims with the tools they need to avoid exorbitant medical bills and fight back against unfair charges. Here’s a look back at 2019’s stories.
Loophole Averted After Surprise-Bill Brouhaha In Texas
The Texas Medical Board bowed out of the rule-making process for a new law protecting consumers from surprise medical bills. Advocates hailed the new rules written by the state insurance regulators.
Sen. Grassley Questions UVA Health On Findings From KHN Investigation
A letter from the Senate Finance Committee chairman questions the University of Virginia Health System about its financial assistance policies, billing practices and prices.